As the patents on many drugs, which have been launched in the 1990s, will soon expire, the pharmaceutical industry finds itself at a turning point in its evolution, particularly as far as research and development are concerned. As the pipelines of new products are shrinking, the exposure of many companies is increasing. This will surely hurt revenue in mid and long term. Further pressure comes from an increasingly turbulent economy, shareholders, greater regulatory burden and rallying operating costs, not to mention growing R&D costs. It is the high clinical development costs, in conjunction with shrinking drug discovery rates that are leading to a decline in the productivity in the pharmaceuticals industry. Moreover, during the last decade, R&D productivity has decreased. But even though emerging technologies have enabled companies to develop multiple parallel options, and to test numerous compounds in early stages, such techniques are effective only when there exist databases of candidates as well as drug evaluation criteria. An important improvement is expected in establishing new methods for identifying unwanted toxic effects in early development phases, as well as reducing the late-stage failure rate. Bioinformatics and biomarkers are expected to play an important role.
However, independently of new technologies, and in order to adapt, the pharmaceutical industry must re-think its current business model which appears to be unsustainable in a rapidly changing and demanding market. Innovative medicines will be in demand, as the need for more personalized treatment grows for an quickly growing and fragmented population. In fact, as diagnosis methods improve, the need for more personalized and focused drugs will be inevitable. Pharmaceutical companies must transition from the old block-buster model to a more fragmented and diversified offering of products. It appears, therefore, that the economical sustainability of the pharmaceutical industry hinges on innovation.
A major concern shared by all drug manufacturers is that of drug toxicity. A candidate molecule under investigation must be validated on animals before authorization for trials in humans is granted. If these preclinical studies show good results, clinical trials with healthy volunteers follow. These have the scope of studying drug efficacy and excluding the presence of toxic effects. Following numerous trials on patients with the targeted disease provide a statistical description of the drug efficacy. There exist essentially two approaches to drug toxicity determination: knowledge-based and the QSAR (Quantitative Structure Activity Relationship) rule-based models, which relate variations in biological activity and molecular descriptors. Evidently, any expert of rule-based system will see its efficacy bounded by the quality and relevance of the employed rules. Because of the inability to predict successfully drug toxicity, drug manufacturers report billion-dollar losses every year.
We wish to formulate a conjecture in relation to drug toxicity: the toxic effects of a molecule are proportional to its complexity. In other words, we suggest that a more complex molecule has greater potential to do damage and over a broader spectrum and that higher complexity may also imply greater capacity to combine with other molecules. The underlying idea is to use complexity as a ranking and risk-stratification mechanism for molecules.
Over the last decade, Ontonix has been developing and validating a novel approach to measuring complexity. The metric is function of structure, entropy, data granularity and coarse-graining. It has been used successfully as an innovative risk-stratification and crisis-anticipation system in economics, medicine and engineering. The metric possesses the following properties:
- The existence of a lower and upper bound. The upper bound is known as critical complexity.
- In the vicinity of its lower complexity bound, a generic dynamic system behaves in deterministic fashion.
- In the vicinity of its critical complexity, a system possesses a very high number of potential behavioral modes and spontaneous mode-switching occurs even in the presence of injection of very small amounts of energy.
- A large number of components is not necessary to lead to high complexity. Systems with a large number of components can be considerably less complex than systems with a very small number of components. In essence, complicated does not necessarily imply complex.
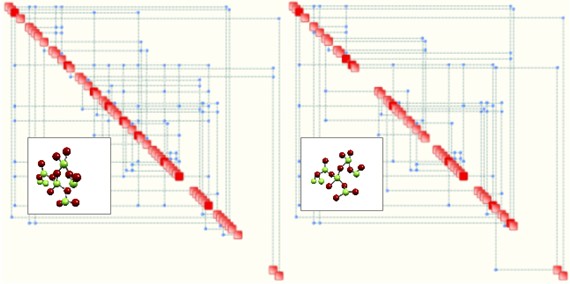
Based on molecular modelling and molecular simulation techniques (Monte Carlo Simulation), one may readily measure the complexity of compounds and use this measure to classify and rank them. In other words, we suggest to use complexity as a “biomarker”. A simple example of the concept is illustrated below, where two so-called Process Maps are shown. Each map is determined by OntoSpace™, a complexity management system developed by Ontonix. Such maps represent the structural properties of a given system, whereby relevant parameters are aligned along the diagonal and are linked by means of connectors (blue dots) which correspond to significant rules. Critical parameters – shown in red – correspond to hubs. The map on the left corresponds to a system with 94 rules and has a complexity of 28.4. The one on the right exhibits 69 rules and a complexity of 19.2. Supposing that both maps correspond to two candidate drugs for the same target disease, the one of the right could correspond to a potentially less toxic candidate.
Clearly, the logic is that if one can perform a given task with a less complex solution that is probably a better solution. However, we also suggest that substances which function in the proximity of their corresponding critical complexities are globally less robust and, potentially, more toxic. Therefore, a higher value of complexity does not necessarily imply a worse alternative – it is ultimately the relative distance to the corresponding critical complexity which may turn out to be a better discriminant.
www.ontomeds.com www.ontonix.com


0 comments on “Complexity-Based Estimation of Drug Toxicity”