One of the most frequent misconceptions about complexity is that the more components a system has the more complex it is. Conversely, a system with less components is bound to be simpler. This may be true, but it is not a general rule.
Let’s take a look at the formal definition of complexity:
C=f(N, S; E)
where N is the number of components (or agents), S is a matrix representing structure and E is the entropy matrix. The “f” in the equation is a spectral operator. C stands for complexity and it is a scalar. The units of complexity are bits, which means that complexity is a measure of structured information.
According to the above equation, the number of components N is not sufficient to define complexity, which is an interplay of Structure and Entropy and Structure-Entropy interactions. Let’s consider, for example, molecules and their complexity.
Chemical molecules are ensembles of atoms, some contain very few, some very many. Three examples (in parentheses numbers of atoms):
Adrenaline (26)

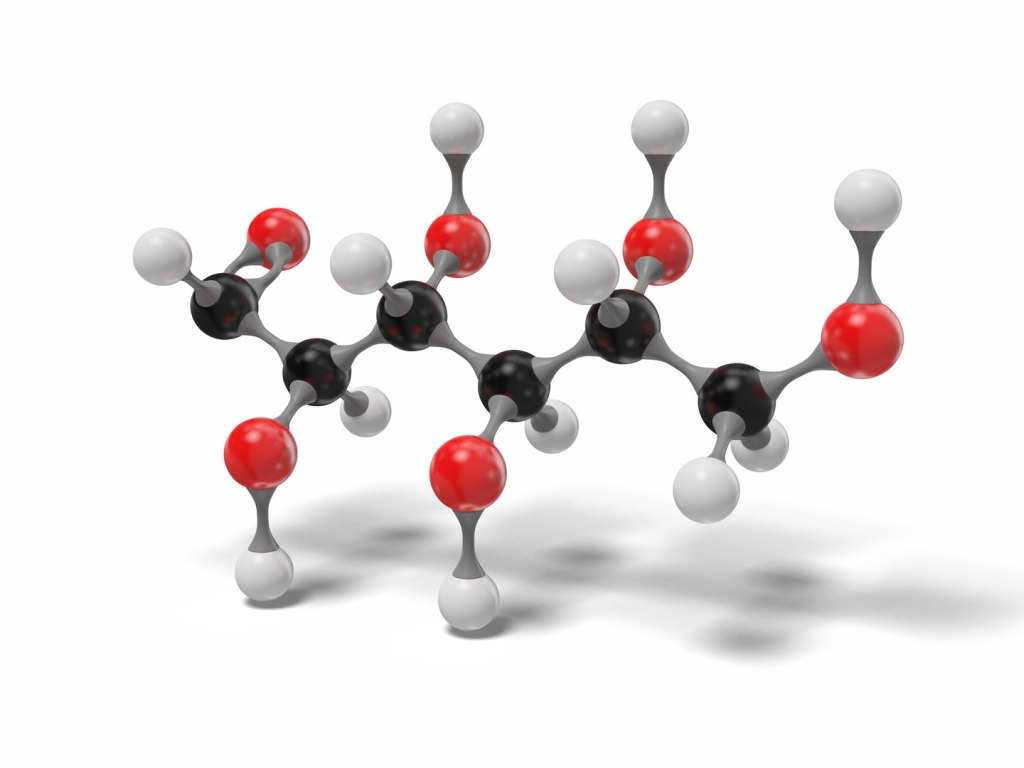
Glucose (24)
Insulin (777)

Adrenaline and glucose have a very similar number of atoms, 26 versus 24, while insulin has 777. There is a high chance that insulin is a more complex molecule than the other two, but it is not easy to confirm. This is why.
In order to build the Structure and Entropy matrices that are needed to measure complexity one must actually see how each atom vibrates and how this vibration relates to the vibrations in all the other atoms. Some atoms may vibrate in unison (in phase), others in opposition, others still in a totally unrelated manner. These vibration patterns are called modes of vibration and the more atoms there are, the more modes of vibration one can identify. Any shape of a vibrating system can be seen as a sum (superposition) of its modes of vibration. Not all modes need to participate in a vibrating system at a given time – it depends on how the system is excited.
An example of modes and modal superposition is shown below, in which the shape of a vibrating beam is decomposed into its basic modes.

A more complex example – that of the SF6 (sulphur hexafluoride) molecule – is illustrated:

The above image is from a paper, where authors discuss how “The use of ultrafast light pulses to visualize chemical dynamics has advanced our fundamental understanding of chemical and biochemical processes, by providing a means of monitoring the motion of atoms within a molecule in real time” from: https://www.researchgate.net/publication/259325378_Inaugural_Article_by_a_Recently_Elected_Academy_Member
Understanding the various modes of vibration, and their associated natural frequencies, is fundamental towards the understanding of a molecule and in particular its dynamics. It is in fact the dynamics of a molecule that provides an expression of its complexity. A molecule that does not vibrate – all atoms occupy exactly the same positions over time – has zero complexity. It is the modes of a molecule that define what it can do and how it can combine with other molecules.
So, the bottom line is that one needs to have a molecule that vibrates, record the x, y, z positions of all of its atoms over a period of time and run all the data through our QCM algorithm which computes the molecule’s complexity (this can be done easily with Molecular Dynamics Simulation codes and a supercomputer). Only then will one be able to see the impact of N, S and E in the value of complexity. In more precise terms, we are not measuring the complexity of the molecule but that of its dynamics. The same molecule can exhibit totally different behaviour, depending on temperature, or the presence of electromagnetic fields or even other molecules.
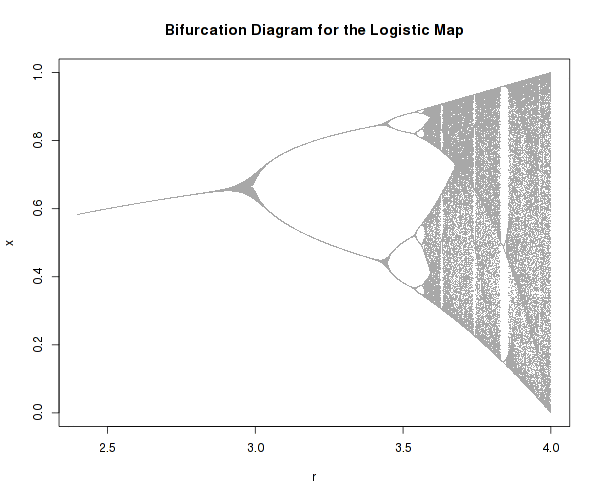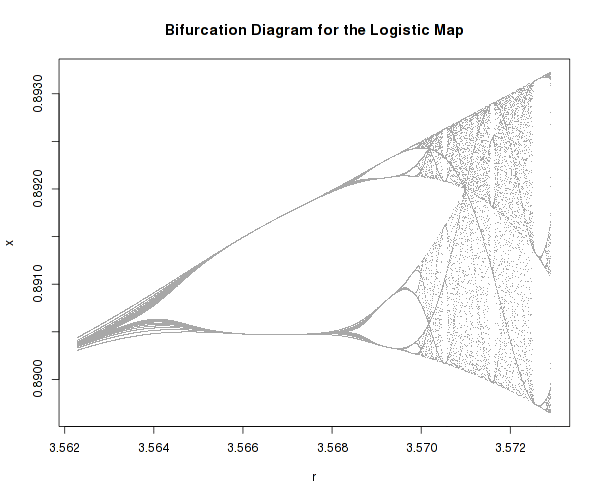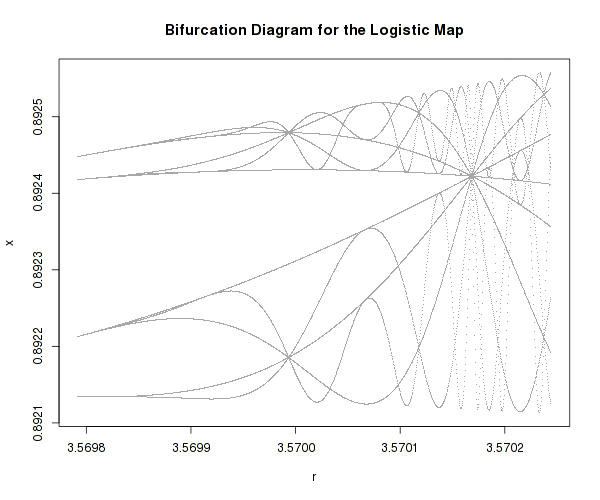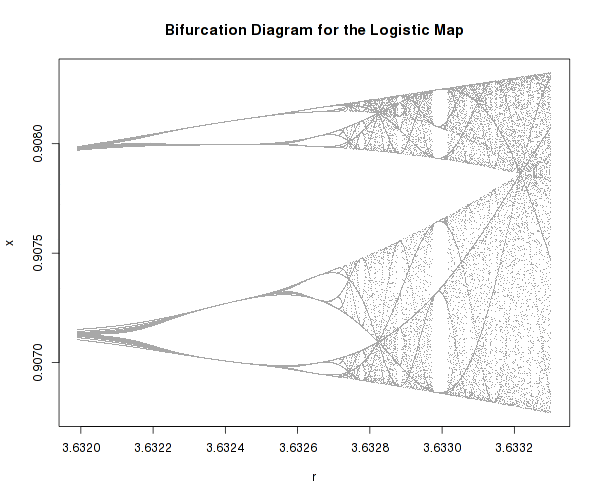
There one well known example of a very simple system that possesses highly complex dynamics is the logistic map:
It is a one-dimensional non-linear equation, with one parameter, r. As one varies r, the above equation can actually develop bifurcations and even chaos as well as self-similarity (fractals).

It is difficult to imagine how such an innocently looking equation can lead to such different and complex dynamic regimes, just because one parameter is changed.
Below we illustrate examples of Complexity Maps, obtained processing Molecular Dynamics simulations of molecules containg, respectively, 10, 30 and 60 atoms. Since for each atom there are three coordinates, the said maps contain, 30, 90 and 180 dimensions.
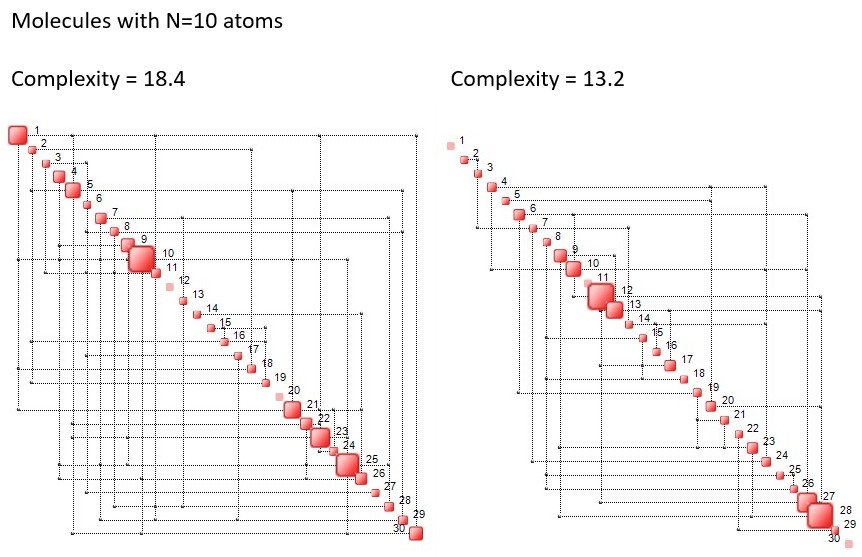


The larger squares on the diagonal correspond to atoms and their corresponding vibration directions which have a large impact on the complexity of the molecule, hence on its information content. In the example below (N=10 atoms) we observe that the vibration of atom 7 in the z direction, that of atom 2 in the x direction and atom 6 in the x direction, contribute over 32% of the molecule’s complexity.

The bar chart on the right is known as the Complexity Profile, or Complexity Spectrum, and it ranks components based on their complexity footprint in the molecule. It may be the case that components at the bottom of the chart indicate which bonds may brake when temperature is increased or when the molecule is exposed to radiation, etc.
Complexity Maps are of paramount importance since they reflect the structure of interactions and information flow/exchange within a system. One of the shortcomings of physics is that while it often addresses structure, it neglects information. To understand a system, any kind of system, is equivalent to the understanding of the structure of the interactions taking place therein and being able to explain them. Our QCT – Quantitative Complexity Theory – establishes a bridge between physics and information. Most physical processes involve some kind of structural transformations and this is why Complexity Maps are so important.
The lesson here is that systems that “look simple” can behave in extremely complex ways. Systems that have a small number of components (e.g., atoms) may exhibit astonishingly complex dynamics. Trying to infer complexity by relying on intuition or counting atoms is risky. Complexity is one form of expression of the dynamics of a system and of its properties. It is not something that may be inferred from a static “picture” or a set of rules.

Reblogged this on Calculus of Decay .
LikeLike